
Supervisory institution:
National Science Centre
Project manager:
M.Sc. Eng. Natalia Michalak
Budget:
73 800 PLN
Start date:
29.02.2017
Duration:
24 months
Contract number:
UMO-2016/21/D/ST3/00975
In heterogeneous catalysis, the structure of catalyst’s surface has the biggest impact on the yield of catalytic reaction. In order to design new catalysts and optimize the commonly used ones, it is necessary to perform fundamental studies that will allow to analyze the kinetics of catalytic reactions in relation to the surface structural changes. Such studies are carried out under idealized ultra-high vacuum (UHV) conditions using idealized model systems.
The aim of this project is to prepare model catalysts consisting of ultrathin (0.25-1 nm) iron oxide FeO(111) islands and films on Ru(0001) and to study the catalytic activity of these systems in model carbon monoxide (CO) to carbon dioxide (CO2) catalytic reaction. Discovered in 2008, the high catalytic activity of FeO(111) films grown on Pt(111) [Sun et al., J. Catal. 266 (2008) 359] started a new wave of studies focused on the investigation of catalytic activity of ultrathin oxide films grown on metal single crystal supports. The activity of FeO(111)/Pt(111) is being mainly attributed to the oxide film-metal substrate interaction. In addition, recent studies highlight the activity of iron oxide islands’ edges in CO oxidation even at room temperature [Fu et al., Science 328 (2010) 1141]. Using Ru(0001), a single crystalline support with different than Pt(111) physico-chemical properties (surface lattice constant, work function), may significantly influence the catalytic activity of FeO.
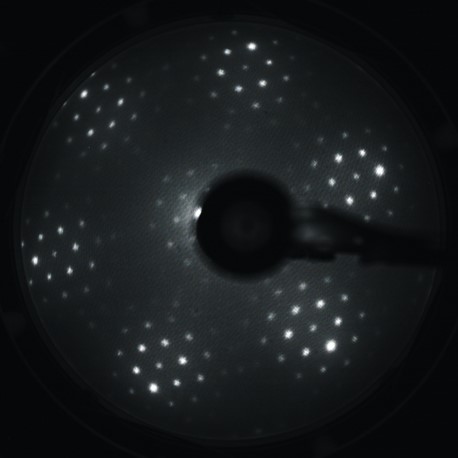
The growth of iron oxide films will be performed by deposition of iron onto the surface of Ru(0001) and subsequent oxidation in molecular oxygen at high temperatures. The structure of fabricated films will be studied using scanning tunneling microscopy (STM), low energy electron diffraction (LEED) and x-ray photoelectron spectroscopy (XPS). As prepared structures will be subjected to CO oxidation reaction in a high pressure cell connected to the main UHV chamber. The course of the reaction will be investigate in situ using gas chromatography (GC). Then, STM, LEED and XPS will be used to study ex situ the structural changes that occur at the catalyst’s surface during the reaction. The kinetics of elementary steps (adsorption/desorption of CO, O2 and CO2) will be analyzed using temperature programmed desorption (TPD).


