Supervisory institution:
National Science Centre
Project manager:
Dr. Paulina Krystyna Skupin-Mrugalska
Budget:
847 150,00 PLN
Project objectives/Hipothesis. The main objective of proposed research project is evaluation of magnetic and relaxation properties - for magnetic resonance imaging (MRI), and anticancer activity - for photodynamic therapy (PDT), of novel theranostic liposomal platforms GdLip, using in vitro cellular and in vivo animal model. Applicants have suggested GdLip liposomes (LIP) as a multifunctional agent for cancer imaging and therapy. A contrast agent (lipid derivative of gadolinium(III) complex salt, Gd-DTPA) and a model photosensitizer (zinc phthalocyanine, ZnPc) will be incorporated into lipid membrane. Different types of LIP will be prepared involving classic, pegylated LIP (long-circulating vehicles, coupled with polyethylene glycol chains) and their actively targeted analogues with epidermal growth factor receptor (EGFR) antibody coupled on the LIP surface. Applicants have assumed that physico-chemical character, composition of lipid bilayer and properties of ZnPc may influence each other and further alter relaxation properties of paramagnetic Gd(III) ion in MR imaging. The hypothesis of the project is that: 1) molar ratio of ZnPc and Gd-DTPA incorporated into GdLip modifies relaxation time of Gd(III) and thus changes relaxivity and MR image contrast; 2) the presence and concentration of Gd(III) ions do not influence cytotoxicity of ZnPc, 3) actively targeted LIP enhance delivery of ZnPc to the targeted cells; 4) GdLip possess relaxation properties comparable with commercial contrast agents such as Magnevist®. Furthermore, it is expected that GdLip will provide a diagnostic and therapeutic option, enabling simultaneous imaging and treatment of tumors overexpressing EGFR receptor, and may provide an opportunity to monitor therapeutic outcomes.
Structure of GdLip "theranostic" platform - a diagnostic-therapeutic agent for simultaneous delivery of antibody, contrast agent and drag subsance
Classic and pegylated liposomes will be obtained by a hydration method. Actively targeted liposomes will be prepared by a conjugation of classic and pegylated ones with EGFR antibody through heterobifunctional cross linker. Quantitative analysis of liposome composition will be assessed by chromatographic method (HPLC). Diameter size and size distribution will be determined by nanoparticle tracking analysis. Transmission electron microscopy will be employed to confirm the determined size of liposomes, study their morphology and distribution of different components within lipid bilayer. The interaction between GdLip and model cellular membrane will be studied by fluorescence correlation spectroscopy. In vitro release profile and kinetics of ZnPc from GdLip will be analyzed by means of asymmetric field flow field fractionation. Cytotoxicity and photocytotoxicity will be evaluated in vitro using human squamous cell carcinoma derived from head and neck cancer and normal fibroblasts, in 2D and 3D cell culture model. Biochemical assays will be used to evaluate cytotoxic effect of GdLip (MTT, LDH), signaling pathways activation (e.g. mediated by EGFR and apoptotic) and to study 3D cell model. Cellular uptake study (ZnPc extraction from cells and determination by HPLC) by cells with diverse expression of
EGFR is expected to provide information on the influence of EGFR antibody decorated LIP on cell ability to actively uptake the GdLip. In vitro cell culture studies will involve also subcellular localization (by confocal microscopy) of ZnPc delivered by GdLip. A standard ZnPc and liposomes containing ZnPc but no Gd(III) ion will serve as a positive control. Magnetic properties and relaxivity of different types of GdLip will be studied in vitro, following cell incubation with liposomes, in magnetic field of different frequencies. Relaxation times will be measured and relaxivity of GdLip will be determined. Imaging of cells labeled with different types of GdLip will be performed using MRI scanner (9.4 T). Parameters describing relaxation properties of GdLip will be compared with literature data of commercially available contrast agent, Magnevist®. Finally, in vivo safety and efficacy study will be performed using mice bearing tumor xenograft model. In vivo research will show the biodistribution of GdLip in organs and tumor, pharmacokinetics and GdLip usefulness for cancer imaging and MR image-guided PDT.
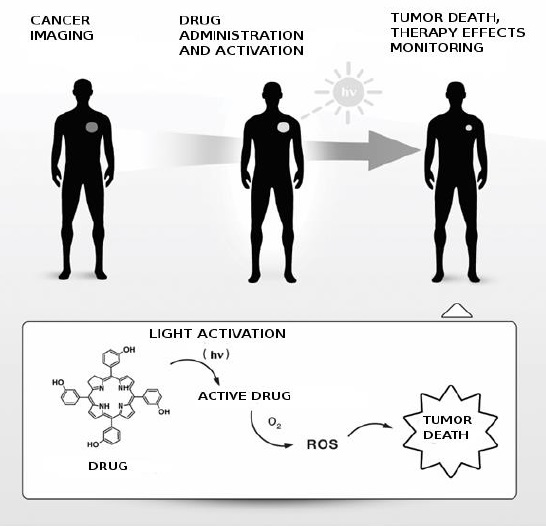
GdLip as a potencial diagnostic and therapeutic agent for cancer imaging and therapy (hv - light, ROS - reactive oxygene species)
Applicants aim to develop a novel strategy enabling MR image guided drug delivery and therapy monitoring in PDT by assessment of in vitro/in vivo properties of multifunctional liposomes GdLip. The concept combines targeted delivery, MR imaging and PDT. In the scientific field, imageguidance and theranostic agents are believed to boost the efficacy of PDT due to the potential of targeted delivery (EGFR antibody) and monitoring the therapy on various levels (Gd-DTPA): before treatment, during drug delivery, release, drug activation and post-treatment. The optimization of GdLip composition may allow to decrease the amount of Gd-DTPA used to obtain a good contrast, because other components of the agent increase the relaxivity. To the best knowledge of applicants, this is the pioneering approach when a liposome-based theranostic agents containing a model photosensitizing structure and lipid derivative of Gd- DTPA are submitted for 2D and 3D in vitro and in vivo studies. The world statistics presented in the GLOBOCAN2012 report, show the permanent increase of the incidence of cancer mortality and morbidity. In the light of the above report, searching and development of novel diagnostic and therapeutic modalities, such as GdLip, seems very important and rational. Furthermore, proposed research tasks employ achievements of nanotechnology and drug delivery (targeted liposomes), advanced analytical techniques and modern methods in toxicology (3D cell culture model). Applicants emphasize that due to participating in such interdisciplinary research project they would have a great opportunity to enhance and diversify research competences. Applicants are convinced that results of the project will be published in highly ranked internationally approved journals.


